|
By Nicholas Raffoul
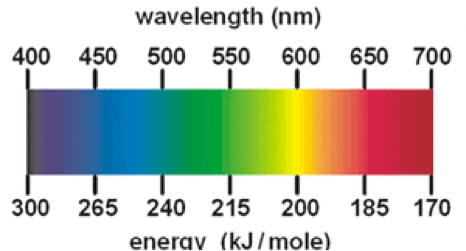
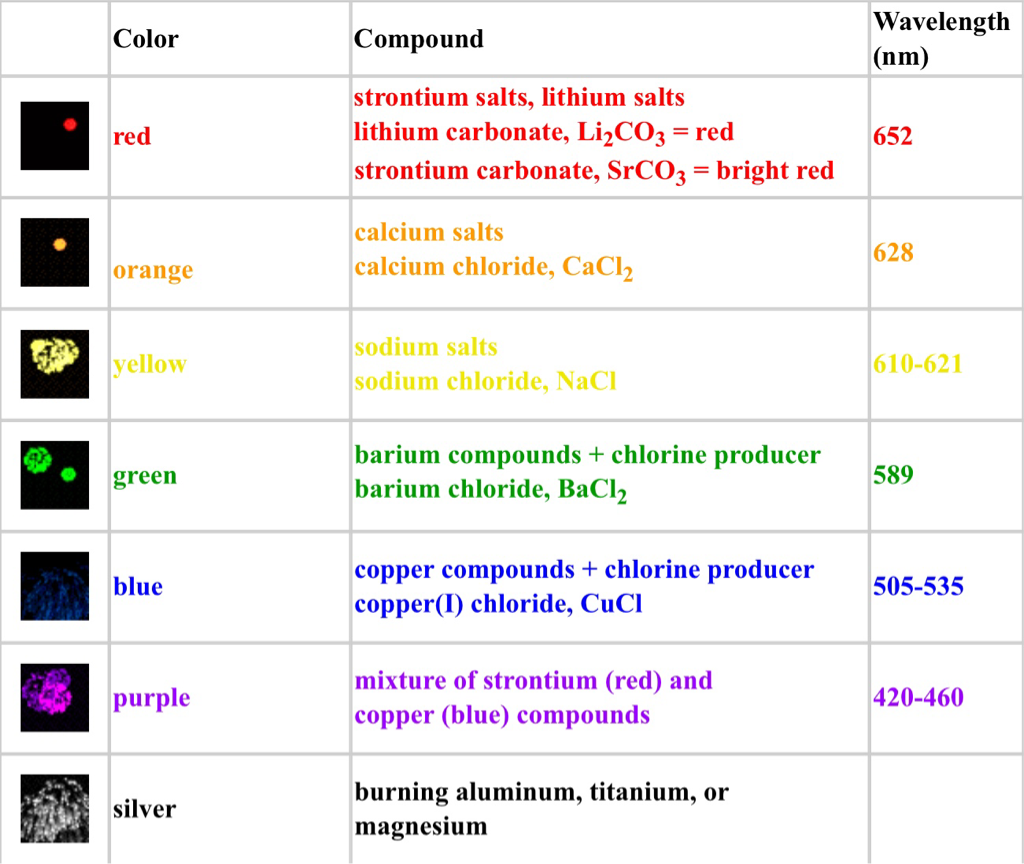
Chemical Changes In Fireworks In a chemical reaction, there is a change in the composition of the substances. As heard of earlier, Fireworks need a source of explodable/combustible material to use for energy. This can be black powder or potassium nitrate. All of the above chemicals help to make a chemical change which makes the amazing, colourful spark, we call fireworks. The chemistry behind fireworks and the chemical changes is that the metals located inside the fireworks are heated up by the flame to the right temperature, the electrons will jump between the energy levels. When they then fall back to a lower state a photon is produced and the different chemicals inside the firework will determine the type of photon/ light colour is emitted. When chemicals are heated, the ions emit characteristic wavelengths or colors of light. This works much like the flame test, a method we used to identify different colours by burning different chemicals. Basically, the chemical change involved in Fireworks is when the black powder or gun powder gets burned and chemically changed into light depending on the chemical mixed with the powder used. The other chemical change is that the shell of the fireworks gets burned into ashes. The table below is to show you which chemicals to mix for which colour you would like to achieve. For example, if you wanted to make a nice red firework you would use strontium and mix it with the black/gun powder. The sights and sounds of each explosion from the fireworks are the result of several chemical reactions. Oxidations and reductions are happening as the firework flys high in the sky. Oxidizers produce the oxygen gas required to burn the mixture of reducing agents and to excite the atoms of the light emitting compounds. Various oxidizers are used in both the black powder and the stars. The most commonly used oxidizers are nitrates, chlorates, and perchlorates. The reducing agents, sulfur and carbon, combine with the oxygen from the oxidizers to produce the energy of the explosion. Oxidisers make the gas needed to burn the mixture of sulphur and carbon (reducing agents) to excite or make the atoms inside the light making compounds (shown in the table to the right). Most oxidizers are used in black powder. Most of the oxidizers are either nitrates, chlorates and perchlorate. The mixture of sulphur and carbon (reducing agents) combine with the oxygen produced by the oxidisers to produce the energy needed to create the explosion for fireworks. Now that we know how the explosion is made, you should also know how much energy is needed to produce the right colour (wavelength). Chemical Element Colour Produced sodium: yellow barium: green strontium: red copper: blue The table to the side is an example of all the chemicals that can make different colours which is their colour, compound and wavelength. Chemical ingredients of fireworks are used to make specific colours. Barium produces green colours when heated, copper salts produces green and blue flames when heated, sodium salts produces a yellow in flame when heated, lithium produces a red colour when heated, magnesium metal produces very bright white light when burned, and strontium produces a bright red colour. This information is shown above in the table. Physical Changes In Fireworks Fireworks are a mixture of different chemical changes but unfortunately there are no physical changes involved in fireworks. In a physical change there is a difference in the appearance, smell, or simple display of a sample of matter without a change in composition. Although there are no Physical changes, talking about physical changes (Definition, more info & examples). Firstly; the best definition of a Physical change is any change not involving a change in the substance and a change that changes physical form. Mainly physical changes can be reversed whereas the chemical changes cannot be reversed. A physical change involves moving between a solid, liquid and gas. Here are some examples: 1.crushing a can 2.melting an ice cube 3.boiling water 4.mixing sand in water 5.breaking a glass 6.dissolving sugar in water 7.shredding paper 8.chopping wood 9.mixing red and green marbles 10.sublimating dry ice |