By Elias Saba
What are the Elements, Mixtures and compounds in fireworks?
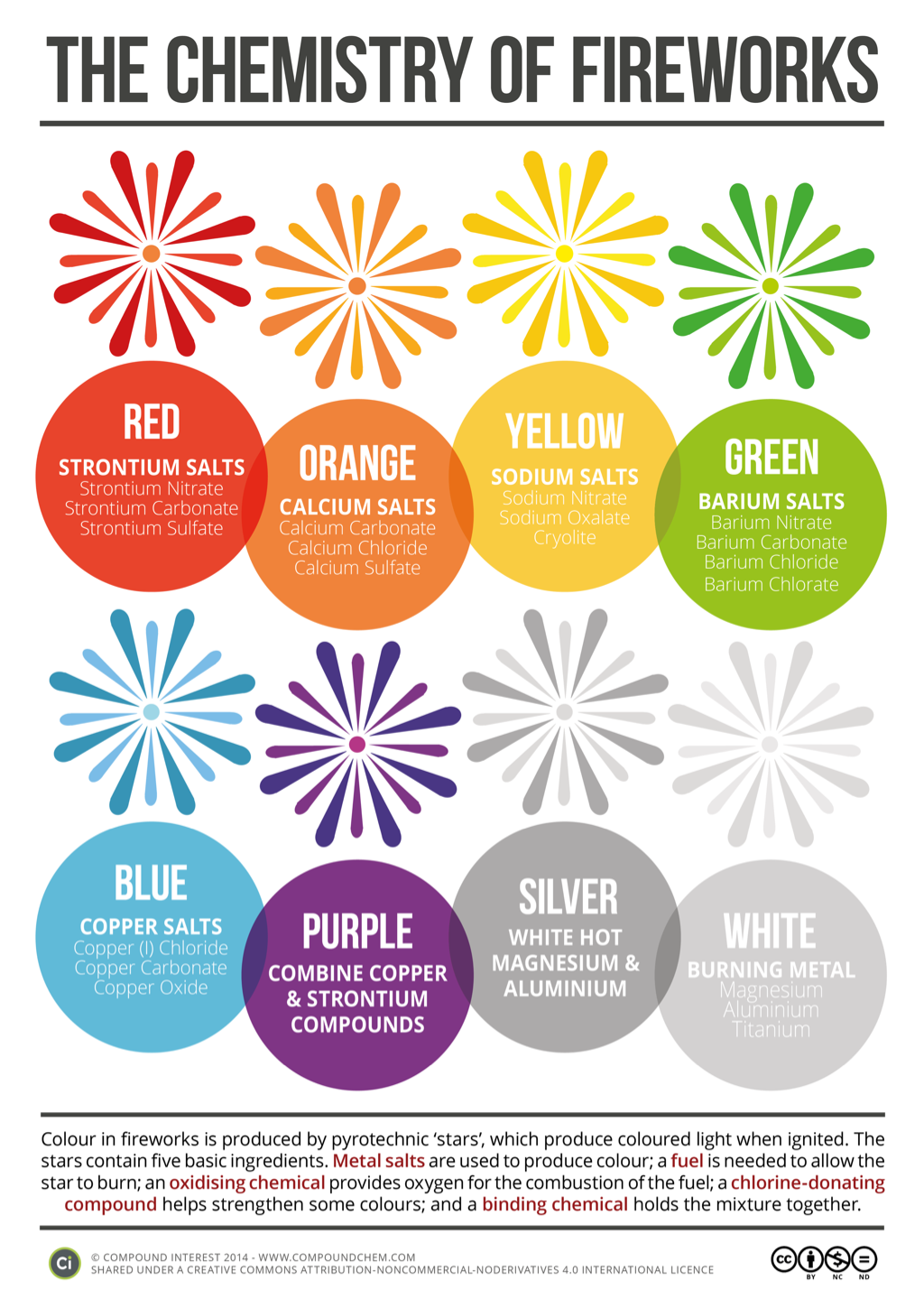
Fireworks need a source of combustable material of energy like black powder, which is made up of a mixture of charcoal sulfur and saltpetre which is another name for potassium nitrate or smokeless powder such as cellulose nitrate. In addition to this fireworks contain substances that give off bright colourful lights when they are heated. A everyday example is if you were to get table salt and throe it into a fire it would light up orange because of the sodium. This is because the electrons in the odium irons are tying to absorb energy and is moving its energy levels higher up and then falling back down to their ground or basis energy levels emitting specific amounts of this energy that are corresponding to the colours of light.
The different chemical elements that create different colours when lighted?
The chemical elements that are chosen to create a firework display are chosen depending on the colour that they want to produce in the firework display. Sodium salts create a colour of yellow as they are heated, Barium compounds produce a colour of green when they are heated, strontium compounds create red colours when they have heated, copper salts also create a blue or green colour when put under heat. The salts contain both metallic cations and non-metallic anions. Anions such as chlorates perchlorates and nitrates these chemicals also produce and contribute to the oxidizing power to the chemical mixture.
Chemical Element.
Colour Produced
sodium yellow
barium green
strontium red
copper Blue
If you would like more information you can visit this website
http://www.chemistryexplained.com/Fe-Ge/Fireworks.html
What are the Elements, Mixtures and compounds in fireworks?
Fireworks need a source of combustable material of energy like black powder, which is made up of a mixture of charcoal sulfur and saltpetre which is another name for potassium nitrate or smokeless powder such as cellulose nitrate. In addition to this fireworks contain substances that give off bright colourful lights when they are heated. A everyday example is if you were to get table salt and throe it into a fire it would light up orange because of the sodium. This is because the electrons in the odium irons are tying to absorb energy and is moving its energy levels higher up and then falling back down to their ground or basis energy levels emitting specific amounts of this energy that are corresponding to the colours of light.
The different chemical elements that create different colours when lighted?
The chemical elements that are chosen to create a firework display are chosen depending on the colour that they want to produce in the firework display. Sodium salts create a colour of yellow as they are heated, Barium compounds produce a colour of green when they are heated, strontium compounds create red colours when they have heated, copper salts also create a blue or green colour when put under heat. The salts contain both metallic cations and non-metallic anions. Anions such as chlorates perchlorates and nitrates these chemicals also produce and contribute to the oxidizing power to the chemical mixture.
Chemical Element.
Colour Produced
sodium yellow
barium green
strontium red
copper Blue
If you would like more information you can visit this website
http://www.chemistryexplained.com/Fe-Ge/Fireworks.html